Where are hydrophobic amino acids found in proteins
Important to tertiary structure are hydrophobic interactions, in which amino. This is a better index of protein hydrophobicity, showing very good correlation with the extent to which residues are buried 5 (correlation coefficient r = 0.9) compared with the hydrophobic indices used previously, and it could be used to characterise tertiary structures. Hydropathy plots cannot identify the membrane-spanning segments of a barrel, as 10 amino acids or fewer are sufficient to traverse a lipid bilayer as an extended strand and only every other amino acid side chain is hydrophobic. interactions between the R groups of the amino acids that make up the protein. Enzymes are proteins that interact in highly regio- and stereo-specific ways with dissolved solutes. The hydrophobic character as defined by the indices given by Tanford 3 and Jones 4 does not reflect hydrophobic environment within protein structures, but we introduce here a new parameter, the ‘bulk hydrophobic character’ obtained from an analysis of the surrounding hydrophobic environment of amino acid residues in protein crystals. During hydrophobic interactions amino acids which are non polar or hydrophobic they align themselves in such a way that all hydrophobic come together and all hydrophilic molecules make hydrogen bonds with water molecules, all hydrophobic amino acid come in to inner side of the protein molecules and formation of the nuclei take place which is. The three-dimensional structure of proteins is a direct consequence of the nature of the covalently-bonded sequence of amino acids, their chemical and physical properties, and the way in which the whole assembly interacts with water. hydrophobic tails only allowing water-based ions to pass via protein channels. In particular the hydrophobic character 2, is a useful parameter in these studies. In saturated fatty acids, all the carbon atoms are connected to other C.
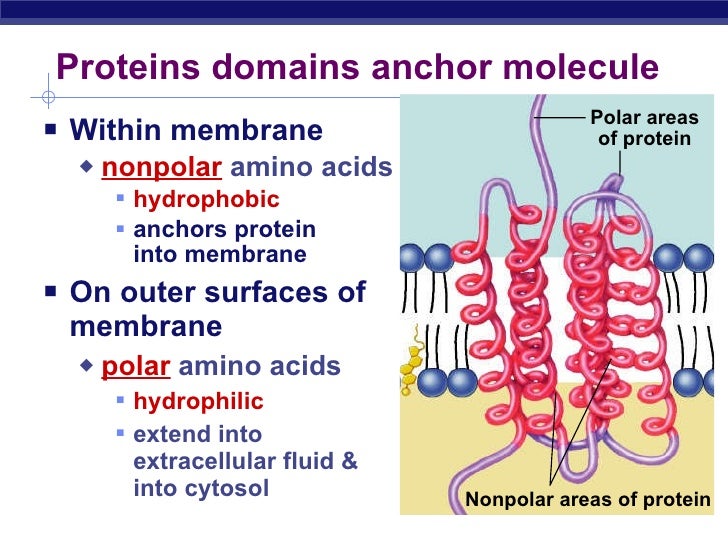
However, the physico-chemical properties 1 of residues can be used to obtain such information. PREDICTIVE studies on the secondary structure of globular proteins aimed at locating ordered structural segments have provided little information about spatial orientations or even as to whether residues are exposed or buried.
